Introduction
The medical device industry is changing fast, right? Companies are always on the lookout for ways to boost efficiency and accuracy in what they do. One exciting strategy that’s gaining traction is the use of Configure Price Quote (CPQ) systems. These systems can really streamline quoting processes and lead to better sales outcomes.
But here’s the catch: the success of implementing CPQ hinges on a few key factors. Think about it-establishing clear configuration guidelines, integrating with ERP systems, providing thorough training for sales teams, and keeping those feedback loops going are all crucial.
So, how can organizations tackle these challenges to tap into the full potential of their CPQ systems and see some serious performance improvements? Let’s dive in!
Establish Clear Configuration Guidelines for Medical Devices
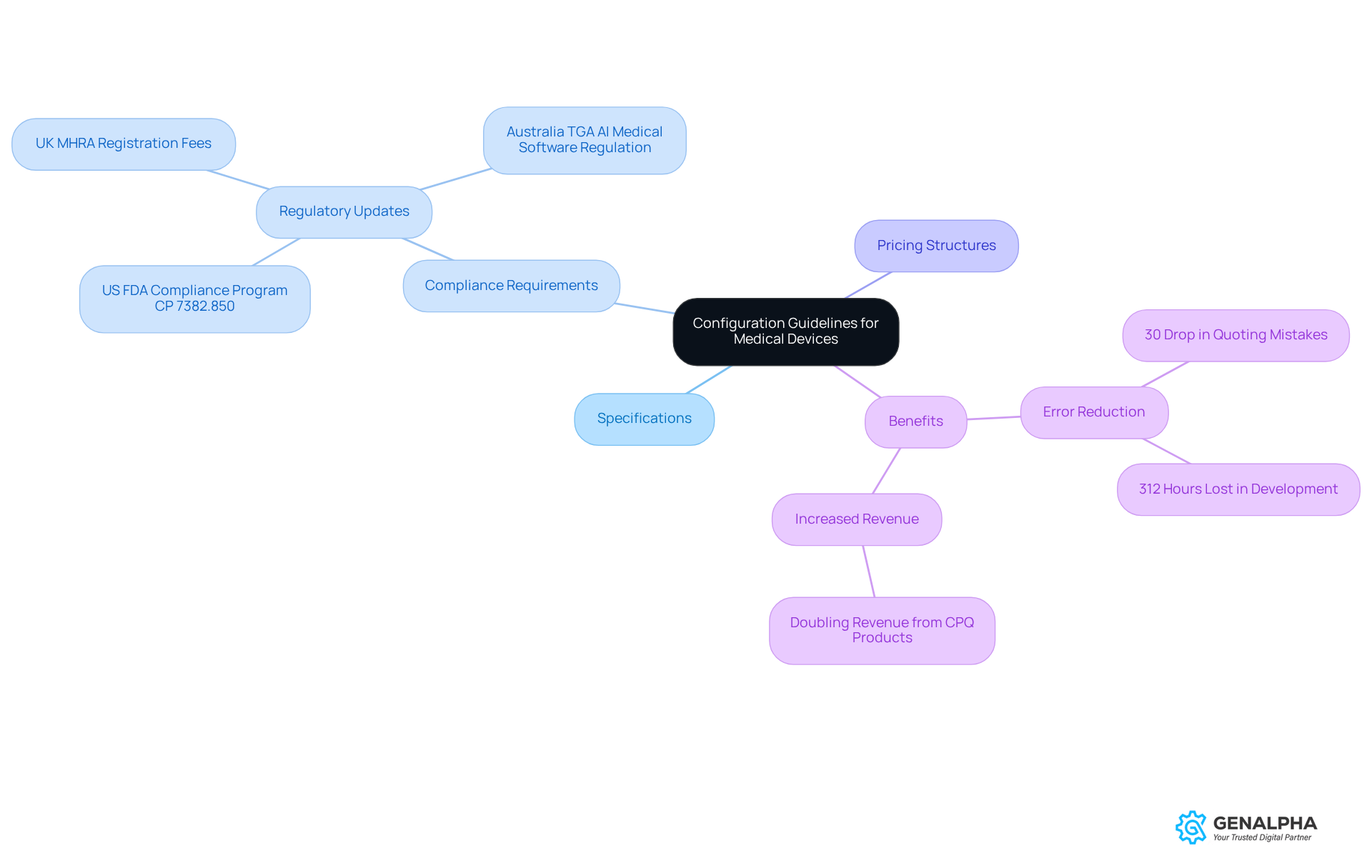
To make sure CPQ implementation in the medical equipment sector goes smoothly, it’s super important to set up clear configuration guidelines. These guidelines should lay out the parameters for product configurations, covering everything from specifications and compliance requirements to pricing structures. By putting together a solid set of rules, organizations can really cut down on errors during the quoting process.
As Jon Speer, a medical equipment expert, puts it, "Quality is an accelerator for leading medical equipment companies." This really highlights how crucial effective guidelines are. For instance, one well-known medical equipment manufacturer created strict configuration protocols that resulted in a 30% drop in quoting mistakes, which significantly boosted their transaction efficiency.
Additionally, a case study showed that another producer doubled its revenue from products supported by CPQ solutions. This just goes to show how effective these systems can be in enhancing performance. And don’t forget, these guidelines should be regularly reviewed and updated to keep up with changes in regulations and product offerings. This way, you ensure ongoing compliance and accuracy. So, how often do you think your organization reviews its guidelines?
Integrate CPQ Systems with ERP Solutions for Seamless Operations
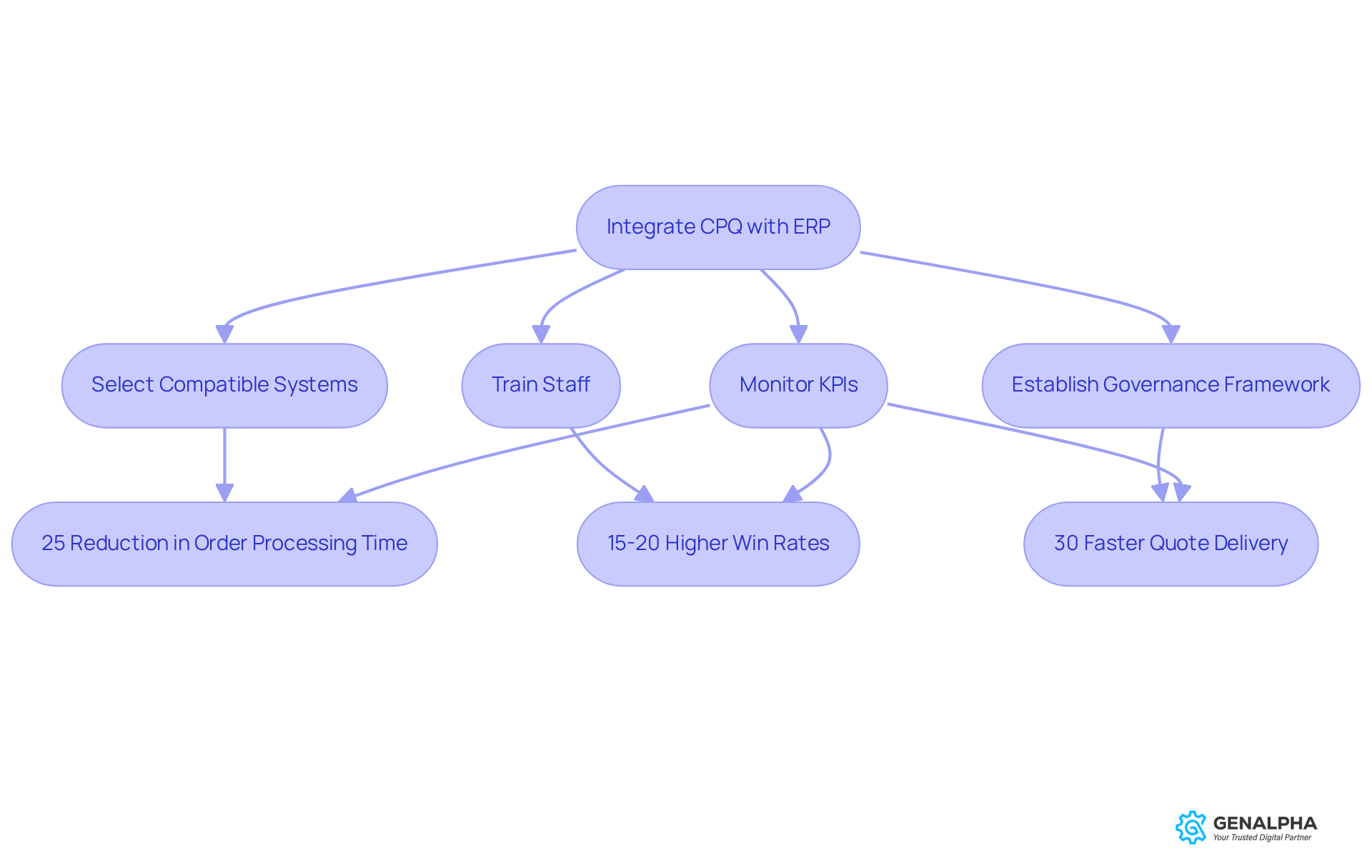
Integrating ERP solutions with medical device CPQ systems is a smart move that can really boost efficiency in medical equipment manufacturing. Imagine this: real-time data sharing through event-driven architectures means everyone on your team has access to the latest info. For example, one medical device company that integrated its medical device CPQ with its ERP system experienced a significant 25% reduction in order processing time and fewer discrepancies between pricing quotes and production orders.
Companies that have these integrated medical device CPQ and ERP setups often report win rates that are 15-20% higher and quote delivery that’s 30% faster. That’s a serious competitive edge! So, how can you achieve this kind of successful integration? Start by picking systems that work well together and invest in training your staff to navigate these interconnected platforms smoothly.
Additionally, setting up governance frameworks to keep an eye on key performance indicators is crucial for long-term success. And let’s not forget about data quality - poor data can really complicate things. Plus, getting accurate configuration validation can lead to a 67% reduction in order modifications after the initial quote is accepted. This really highlights how integration can streamline operations and boost customer satisfaction through accurate and timely order fulfillment.
So, are you ready to take the plunge into integration? It could be the game-changer your organization needs!
Provide Comprehensive Training and Support for Sales Teams
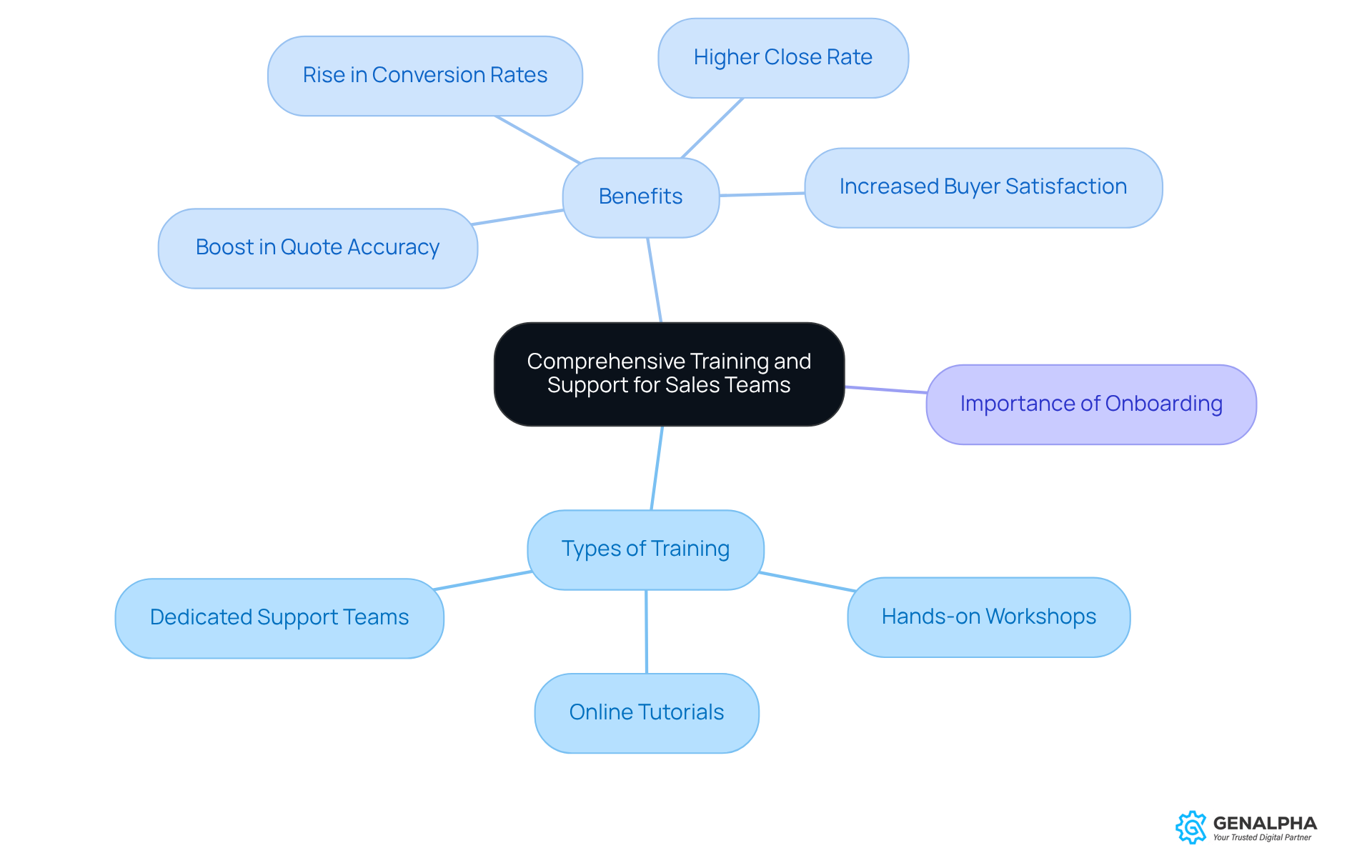
To really make the most of CPQ implementation, organizations need to focus on thorough education and ongoing support for their sales teams. This training should cover not just the technical aspects of the CPQ system but also best practices for engaging with customers and assessing their needs. Think about it: effective development programs often include hands-on workshops, online tutorials, and access to dedicated support teams.
For example, a medical device CPQ company that invested in comprehensive training for its sales staff saw a remarkable 40% boost in quote accuracy and a 20% rise in conversion rates. Plus, coaching can lead to a 41% higher close rate and a 29% increase in buyer satisfaction for virtually trained reps. Continuous support and refresher courses are crucial for helping the team adapt to updates and new features in the CPQ system, ensuring they stay successful in a competitive market.
And let’s not forget about organized onboarding programs - they can significantly enhance the effectiveness of educational initiatives and contribute to long-term success. With the global training market expected to grow to nearly $19 billion by 2032, investing in comprehensive education is more important than ever. So, what are you waiting for? Let's prioritize training and support to empower our sales teams!
Implement Continuous Feedback Loops for Ongoing Improvement
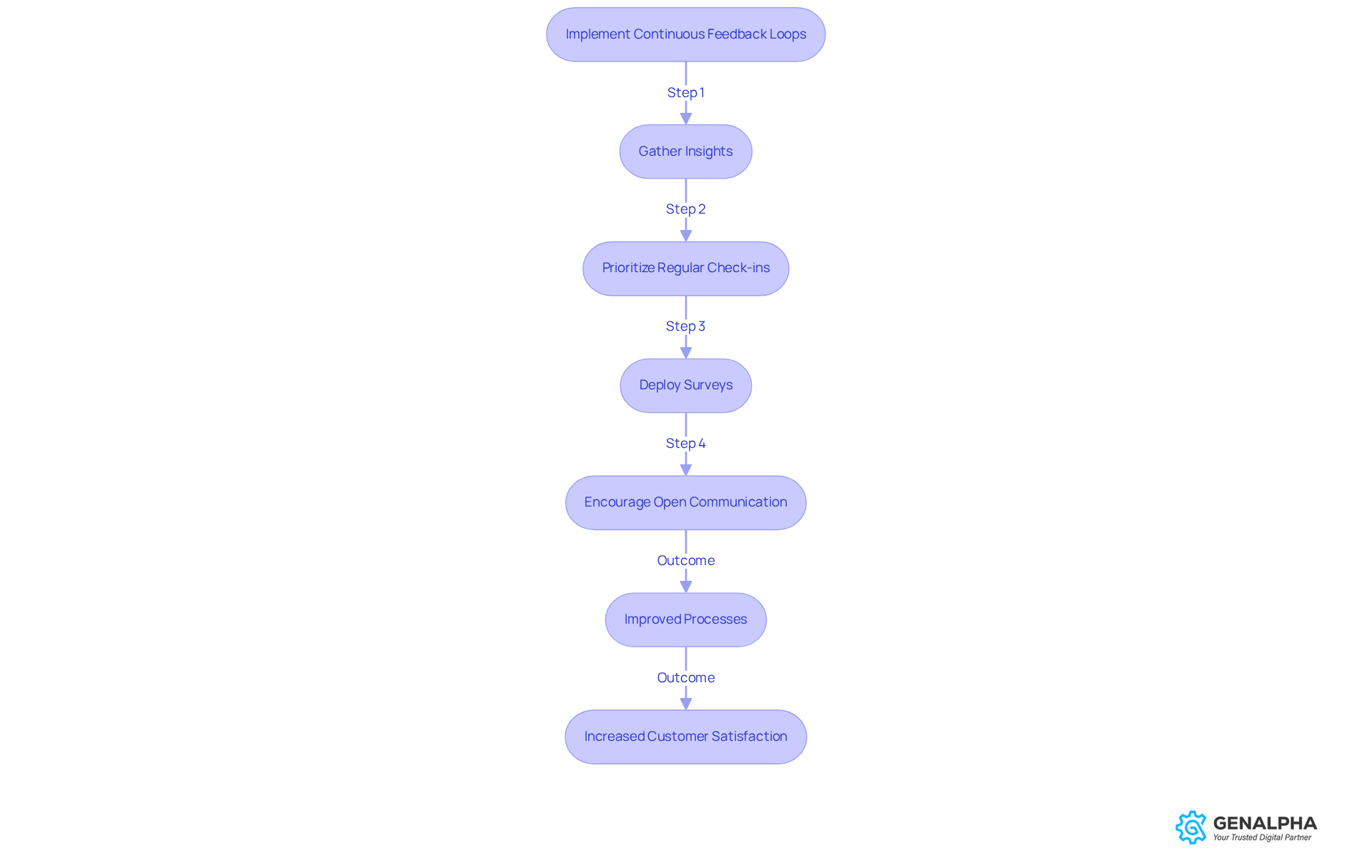
You know, implementing continuous feedback loops is super important for refining processes in medical device cpq for medical equipment companies. By regularly gathering insights from marketing teams and customers, organizations can really pinpoint those pain points and spot opportunities for improvement. For example, one medical device manufacturer set up a feedback loop and saw a fantastic 15% boost in customer satisfaction scores after making some targeted changes based on sales team feedback.
So, how can companies foster effective feedback loops? Well, it’s all about prioritizing regular check-ins, deploying surveys, and encouraging open communication across teams. This iterative strategy not only helps the medical device cpq system adapt to real-world challenges but also aligns it more closely with customer expectations. In the end, this drives improved satisfaction and operational efficiency.
Isn’t it exciting to think about how simple adjustments can lead to such significant results? Let’s keep the conversation going and explore how we can implement these strategies in our own organizations!
Conclusion
When it comes to implementing CPQ in the medical device sector, establishing best practices is key for organizations looking to boost efficiency, accuracy, and overall performance. Think about it: by focusing on clear configuration guidelines, integrating CPQ systems with ERP solutions, providing thorough training for sales teams, and setting up continuous feedback loops, companies can really enhance their quoting processes and ramp up customer satisfaction.
Let’s break it down. First off, having structured configuration guidelines helps minimize errors and ensures compliance. Plus, when CPQ integrates with ERP systems, it allows for real-time data sharing, which means faster order processing and improved accuracy. And don’t forget about training! Investing in comprehensive training for sales teams not only sharpens quote accuracy but also boosts conversion rates. Finally, fostering those continuous feedback loops lets organizations adapt and refine their processes, aligning them more closely with what customers truly need.
So, what’s the takeaway? The successful implementation of CPQ systems in medical devices really hinges on a multifaceted approach that prioritizes clarity, integration, education, and feedback. As the industry evolves, embracing these best practices will be crucial for keeping a competitive edge and driving sustained growth. We encourage organizations to take a good look at their current strategies and think about how these insights can spark innovation and excellence in their operations.
Frequently Asked Questions
What are configuration guidelines for medical devices?
Configuration guidelines for medical devices are rules that outline the parameters for product configurations, including specifications, compliance requirements, and pricing structures.
Why are clear configuration guidelines important in the medical equipment sector?
Clear configuration guidelines are important because they help reduce errors during the quoting process, ensuring smoother CPQ (Configure, Price, Quote) implementation.
How do effective configuration guidelines impact quoting mistakes?
Effective configuration guidelines can significantly lower quoting mistakes; for example, one medical equipment manufacturer saw a 30% drop in quoting errors after implementing strict configuration protocols.
What benefits can organizations expect from using CPQ solutions?
Organizations can expect enhanced performance, as evidenced by a case study where a producer doubled its revenue from products supported by CPQ solutions.
How often should configuration guidelines be reviewed and updated?
Configuration guidelines should be regularly reviewed and updated to keep up with changes in regulations and product offerings, ensuring ongoing compliance and accuracy.




